We offer this product from a manufacturer in India, a preferred vendor for many complex molecules requiring multistage synthesis.
Manus Aktteva Biopharma LLP
If you need a partner for Active Pharmaceutical Ingredients supply from India, Manus Aktteva Biopharma LLP is here to help. We can help our partners obtain APIs for their Research and product development needs, and we serve over 50 countries from our central headquarters in Gujarat, India. Learn more about our APIs below, or enquire now for more information.
What Is An API?
An API, or Active Pharmaceutical Ingredient, is the biologically active ingredient in a particular drug or the finished formulation that causes its effects.
For example, a painkiller will contain active ingredients that suppress pain, but these are present in only very small amounts. APIs are listed on the outside of product packaging, complete with information about the amount contained in each type of medicine.
API or Active Pharmaceutical Ingredients manufacturing pharma companies use these ingredients, combined with inactive ingredients in pills, liquids, or other forms of medicine delivery, to create their generic products or finished formulations.
It’s important to note that these are distinct from raw materials in pharmaceutical manufacturing. Active Pharmaceutical Ingredients – APIs are the result of combining multiple chemical intermediate raw materials, which are the building blocks of the ingredient. Raw materials, in and of themselves, do not have the same effects as an API.
We Source and Deliver High-Quality Active Pharmaceutical Ingredients – APIs from Our Manufacturer Suppliers.
If you need active pharmaceutical ingredients for R&D, product development, or commercial requirements, Manus Aktteva Biopharma LLP is the best sourcing nodal worldwide. As a global sourcing company and fulfillment partner, we can assist our clients in obtaining high-quality ingredients for pharmaceutical and nutraceutical products across the globe.
Our active pharmaceutical ingredient manufacturer suppliers are dedicated to quality. All of the ingredients from our active pharmaceutical ingredient manufacturers are not only ISO-certified, but also meet CGMP, GMP, and US FDA or EUGMP requirements for quality and integrity.
We invite potential customers to contact us right away to learn more about our products, and to get samples of the active pharmaceutical ingredients and pharmaceutical intermediates offered by Manus Aktteva Biopharma LLP. We guarantee that our product samples will meet or exceed your quality requirements.
Contact Us Now For More Information About Our Products & Services
Working with reputable pharmaceutical API manufacturers ensures the best results when developing, marketing, and manufacturing a new drug or related product. If you wish to get the active ingredients you need at a low cost – without compromising quality – Manus Aktteva Biopharma LLP is the best choice. To get free samples, learn more about our manufacturing and sourcing solutions, or place an order, please contact us online. We are always here to help our customers get the chemicals and pharmaceutical ingredients they need – worldwide.
-

We offer this product from a manufacturer in India having ISO 9001: 2008, ISO 14001, ISO 18001 and are a WHO - GMP Certified Company.
As a Pharmaceutical company, the factory manufactures all the APIs developed in their own R&D. Currently, and the R&D is involved in developing a few more API products. We have a Biotech drug discovery R&D jointly developed with an Israeli partner to produce Recombinant fusion proteins to treat and cure asthma and Mastocytosis.
-
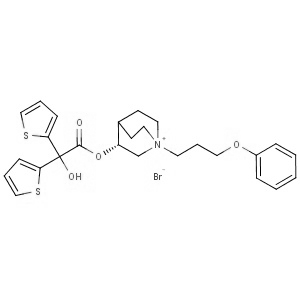
We offer from our manufacturer principals facility located in India with the SOP driven manufacturing activity carried out strictly as per ICH Q7 cGMP guidelines, and several conventional and modern techniques are used for the isolation, semi-synthesis, and purification of our products. The manufacturing processes have been validated to ensure consistent product quality.
Facilities have been inspected and approved by Regulatory authorities like US FDA, ANSM, KFDA, TGA, CDSCO, etc. They hold GMP certificates and Written Confirmation for most of our products. They have PMDA accreditation for several products
-
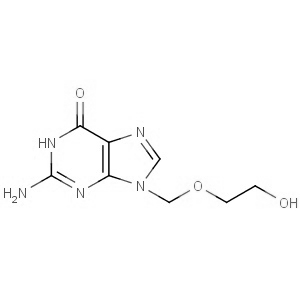 We are offering the product from our Indian manufacturer's USFDA, Japan PMDA, Korea FDA approved site. Their facility is COFEPRIS inspected for several products. Their R&D facility is recognized by the Department of Scientific and Industrial Research (DSIR, Ministry of Science and Technology, Government of India).
We are offering the product from our Indian manufacturer's USFDA, Japan PMDA, Korea FDA approved site. Their facility is COFEPRIS inspected for several products. Their R&D facility is recognized by the Department of Scientific and Industrial Research (DSIR, Ministry of Science and Technology, Government of India). -
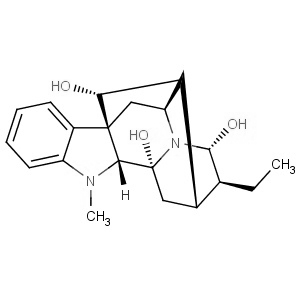 We offer from our manufacturer principals facility located in India with the SOP driven manufacturing activity carried out strictly as per ICH Q7 cGMP guidelines, and several conventional and modern techniques are used for the isolation, semi-synthesis, and purification of our products. The manufacturing processes have been validated to ensure consistent product quality. Facilities have been inspected and approved by Regulatory authorities like US FDA, ANSM, KFDA, TGA, CDSCO, etc. They hold GMP certificates and Written Confirmation for most of our products. They have PMDA accreditation for several products
We offer from our manufacturer principals facility located in India with the SOP driven manufacturing activity carried out strictly as per ICH Q7 cGMP guidelines, and several conventional and modern techniques are used for the isolation, semi-synthesis, and purification of our products. The manufacturing processes have been validated to ensure consistent product quality. Facilities have been inspected and approved by Regulatory authorities like US FDA, ANSM, KFDA, TGA, CDSCO, etc. They hold GMP certificates and Written Confirmation for most of our products. They have PMDA accreditation for several products -
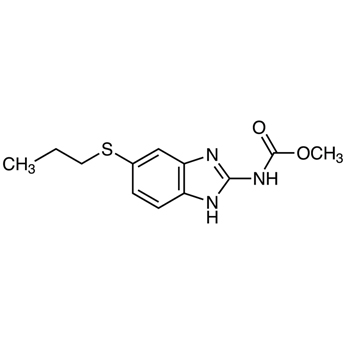 We are offering the product from our Indian manufacturer's USFDA, Japan PMDA, Korea FDA approved site. Their facility is COFEPRIS inspected for several products. Their R&D facility is recognized by the Department of Scientific and Industrial Research (DSIR, Ministry of Science and Technology, Government of India).
We are offering the product from our Indian manufacturer's USFDA, Japan PMDA, Korea FDA approved site. Their facility is COFEPRIS inspected for several products. Their R&D facility is recognized by the Department of Scientific and Industrial Research (DSIR, Ministry of Science and Technology, Government of India). -
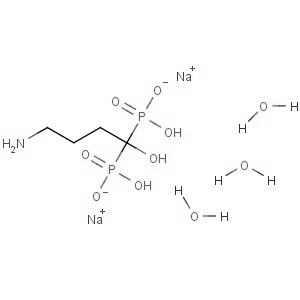
We offer this product from an Indian manufacturer having two-world class manufacturing sites, USFDA and COFEPRIS approved, WHO GMP certified, and have received AFM certificate from Japanese PMDA and recently concluded Korean FDA audit successfully. Further, the facility is also compliant with all other major regulatory bodies, including UKMHRA, EDQM, and TGA.
-

We offer this product from a manufacturer in India having ISO 9001: 2008, ISO 14001, ISO 18001 and are a WHO - GMP Certified Company.
As a Pharmaceutical company, the factory manufactures all the APIs developed in their own R&D. Currently, and the R&D is involved in developing a few more API products. We have a Biotech drug discovery R&D jointly developed with an Israeli partner to produce Recombinant fusion proteins to treat and cure asthma and Mastocytosis.